Marine Scotland just issued @MowiScotlandLtd with a "LICENCE TO DEPOSIT ANY SUBSTANCE OR OBJECT WITHIN THE SCOTTISH MARINE AREA –MARINE FARM" at Ardnish in Loch Ailort https://t.co/B5JB6Z94Uk Is this to use the toxic neonicotinoid Imidacloprid? #Toxic @WeAreBenchmark @ScottishEPA pic.twitter.com/TZD9jNsgpE
— Don Staniford (@TheGAAIA) June 26, 2020
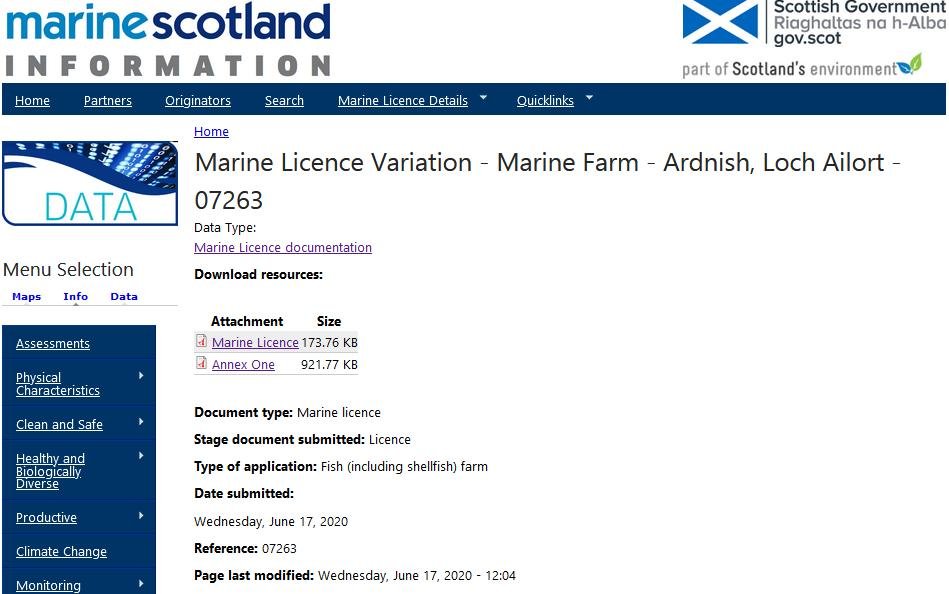
Scottish Salmon Watch stumbled across this interesting 'Marine Licence' issued on 18 June 2020 by the Scottish Government for Mowi's salmon farm at Ardnish in Loch Ailort (the same site at which Mowi is seeking to field trial the toxic neonicotinoid insecticide Imidacloprid).
Here's an excerpt of the 'Licence to Deposit Any Substance or Object Within the Scottish Marine Area – Marine Farm':
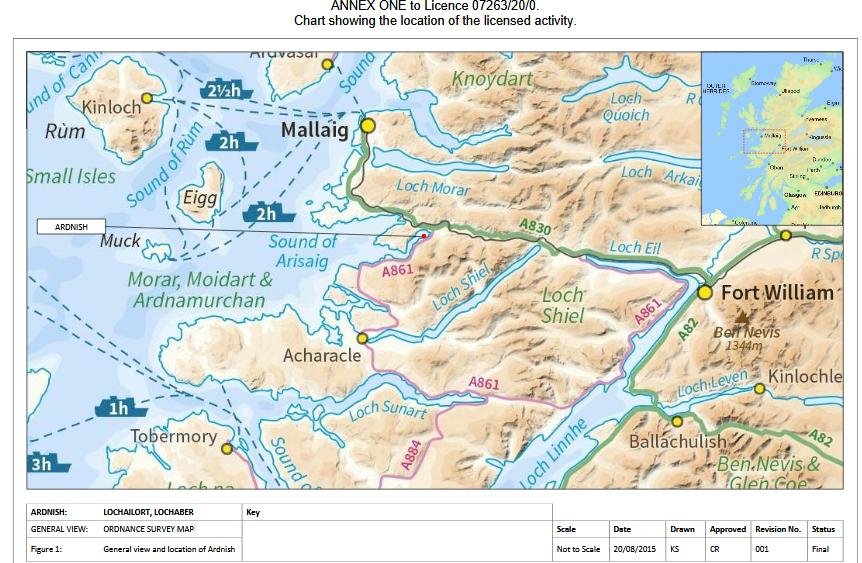
Here's a copy of the map in Annex One:
What does Mowi want to do with 92 cages & 6 mobile pens in Loch Ailort?! https://t.co/0ARGQ1RPSu pic.twitter.com/KI825UyI9J
— Don Staniford (@TheGAAIA) June 30, 2020
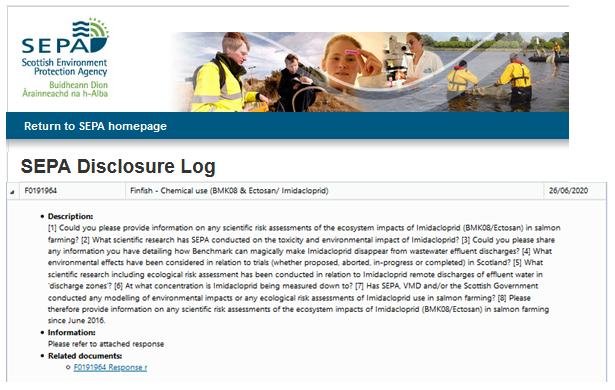
Scottish Salmon Watch emailed SEPA (26 June 2020) asking whether this licence permitted the discharge of Imidicaloprid:
Date: Fri, Jun 26, 2020 at 3:08 PM
Subject: Mowi licence to use Imidacloprid granted on 18 June 2020 in Loch Ailort?
To: Pollard, Peter <peter.pollard@sepa.org.uk>
See: http://marine.gov.scot/ml/marine-licence-marine-farm-ardnish-loch-ailort-07263
In relation to a previous inquiry on Imidacloprid, SEPA told Scottish Salmon Watch (26 June 2020):
From: Pollard, Peter <Peter.Pollard@sepa.org.uk>
Date: Friday, June 26, 2020
Subject: Re: FOI re. Scientific Concerns re. Imidacloprid Use in Scottish Salmon Farming
To: "salmonfarmingkills@gmail.com" <salmonfarmingkills@gmail.com>
Dear Don,
In your email of 8th June requesting access to information held by SEPA, you asked three wider questions. Our answers to these are provided below. You will receive the information we hold relating to the elements of your request that fall under FOI/EIR in separate correspondence.
- Will SEPA be publishing a scientific dossier on environmental impacts of Imidacloprid (as SEPA has done so via Emamectin benzoate)?
SEPA is not planning to commission and publish a scientific dossier on the environmental impacts of imidacloprid.
The 2016 review of emamectin benzoate was prompted by indications from a study by the Scottish Association for Marine Science that the use of the medicine may be affecting crustaceans The purposes of the review were to:
(a) determine if the environmental standards that had been established in 1999 remained scientifically valid; and
(b) potentially derive new standards according to the latest regulatory guidance.
In the case of imidacloprid, there are currently no established coastal water environmental standards to review. There are also no authorised discharges into coastal waters.
Before we would consider authorising any discharges of imidacloprid, we would require the applicant to provide the necessary data to identify a suitable environmental standard in the form of a predicted no effect concentration. It is the responsibility of the applicant to provide the scientific information for this purpose.
Any application made that would result in a breach of an environmental standard would be refused.
- Will SEPA be conducting scientific studies and monitoring of impacts of Imidacloprid (as it has done so via Emamectin benzoate)?
As also noted above, SEPA has not issued any authorisations to any finfish farm-related operation to discharge imidacloprid nor is SEPA in receipt of any application to discharge imidacloprid.
If an application is made to discharge imidacloprid and an authorisation to do so granted, SEPA will carry out appropriately rigorous checks to ensure that the conditions of the authorisation are being met.
The form of the checks will depend on the particular conditions of authorisation, which will in turn depend on the details of the proposed discharge. The checks may include, for example, sampling of the discharge and environmental monitoring.
- Will Bayer/Monsanto be making available to the public their 'Confidential' work on Imidacloprid (or will the chemical company behind Imidacloprid be taking the Merck behind the scenes route)?
We do not have any information on the intentions of Bayer/Monsanto.
Yours sincerely,
Peter
Head of Ecology
Evidence & Flooding
Scottish Environment Protection Agency
A FOI reply from SEPA also dated 26 June 2020 stated:
Read FOI reply from SEPA in full online here
SEPA refuse to disclose information on the use of the toxic neonicotinoid insecticide Imidacloprid in salmon farming citing commercial confidentiality https://t.co/6dqW4x0iel @ScottishEPA @FOIScotland If disclosed it would prejudice economic interests @WeAreBenchmark @FerdOwner pic.twitter.com/m88qS2Tv2A
— Don Staniford (@TheGAAIA) June 30, 2020
The Antithesis of the Precautionary Principle: "SEPA have not conducted any modelling of environmental impacts or ecological risk assessments of Imidacloprid use in salmon farming" https://t.co/6dqW4x0iel @ScottishEPA @WeAreBenchmark @FerdOwner @vmdgovuk @marinescotland #Toxic pic.twitter.com/HCq5kViBxg
— Don Staniford (@TheGAAIA) June 30, 2020
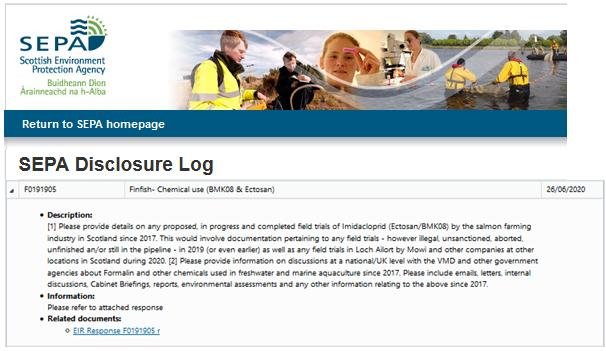
Another FOI reply from SEPA dated 26 June 2020 (FO191905) included:
@NicolaSturgeon stands behind the podium daily spouting about Precautionary and with her full knowledge @strathearnrose @TerryAHearn are allowing poison into the marine environment @marinescotland @ScottishEPA where are u? Suppressed? Silenced https://t.co/2Wb0UUuMQM
— A.L.Paterson (@ALPaterson4) June 30, 2020
Scottish Salmon Watch's letter to SEPA's Chief Executive Terry A'Hearn dated 17 March 2020 (which has still not received a reply over three months later) is available online here
Shame on Sepa and the Scottish Government for protecting commercial confidentiality rather than the environment @ScottishEPA @TerryAHearn @marinescotland @GreenerScotland @FOIScotland @WeAreBenchmark @SSPOsays @FerretScot @ASC_aqua @vmdgovuk @Bayer https://t.co/6u7ZifHNFo
— Don Staniford (@TheGAAIA) March 17, 2020
Read more via:
- BBC Farming Today on "Neonicotinoids in Salmon Farming"
- Sunday Times: "Outlawed insecticide may be used on Scottish salmon"
- Horrible History: Imidacloprid Use in Salmon Farming
- Further Scientific Opposition to Imidacloprid Use in Salmon Farming
- Sunday Times: "Outlawed insecticide may be used on Scottish salmon"
- Scientific Opposition to Neonicotinoid Use in Salmon Farming
- Backgrounder: "Imidacloprid – A Neonicotinoid Insecticide Toxic to Aquatic Life"
- Scottish Greens "Deeply Concerned" at Mowi's Field Trial of Neonicotinoid Imidacloprid in Loch Ailort
- Letter to the Prince of Wales re. bee-killing Imidacloprid use at royal warrant holder Mowi
- Secret Trials: 'Royal' Salmon Doused with Bee-Killing Insecticide Imidacloprid?
- Follow the Salmoney – the Norwegian tobacco billionaire behind the Neonicotinoid Imidacloprid
- The Ferret: "Fish farm companies ‘bidding to use bee-harming pesticide’"
- Revealed: Toxic Neonicotinoid Insecticide Used to 'CleanTreat' Lousy Salmon
Listen to BBC Farming Today on 'Neonicotinoids in Salmon Farming' @BBCFarmingToday https://t.co/CyEaZLuyEg @salmonfarming1 @WeAreBenchmark @MowiScotlandLtd @ScottishEPA @FergusEwingMSP @markruskell @DaveGoulson @MasumiYamamuro @FerdOwner @ASC_aqua @rspcaassured @vmdgovuk pic.twitter.com/aVyi0oD2AH
— Don Staniford (@TheGAAIA) June 16, 2020