Includes:
Read FOI letter in full online here
Read more via:
- Scientific Opposition to Neonicotinoid Use in Salmon Farming
- Backgrounder: "Imidacloprid – A Neonicotinoid Insecticide Toxic to Aquatic Life"
- Secret Trials: 'Royal' Salmon Doused with Bee-Killing Insecticide Imidacloprid?
- Follow the Salmoney – the Norwegian tobacco billionaire behind the Neonicotinoid Imidacloprid
- ENDS Report: "Calls for greater transparency over ‘toxic’ new fish farm product"
- Letter to SEPA: Please Come Clean on Imidacloprid!
- Letter to Scottish Ministers, SEPA & the VMD re. Imidacloprid
- FOI requests on Imidacloprid to SEPA, VMD & the Scottish Government
- Revealed: Toxic Neonicotinoid Insecticide Used to 'CleanTreat' Lousy Salmon
- Please Come Clean on BMK08!
The VMD replied on 6 July 2020:
From: Andrew Saunders <a.saunders@vmd.gov.uk>
Date: Mon, Jul 6, 2020 at 12:48 PM
Subject: Official Sensitive: Response to Freedom of Information Request ATI0662
To: Don Staniford <salmonfarmingkills@gmail.com>
Dear Don,
Thank you for your email dated 8th June 2020.
Your Request
You asked for:
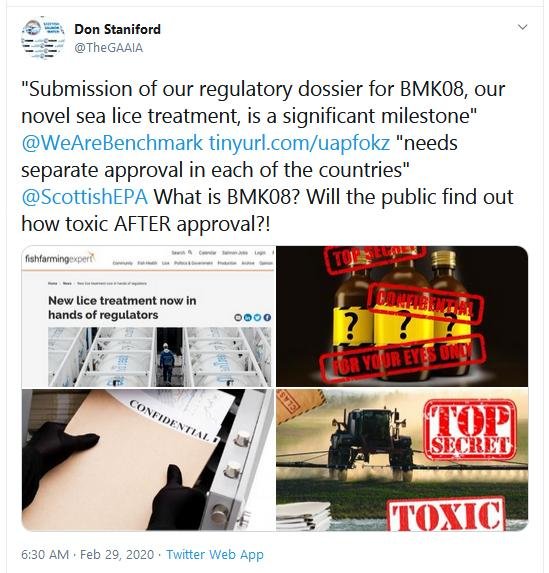
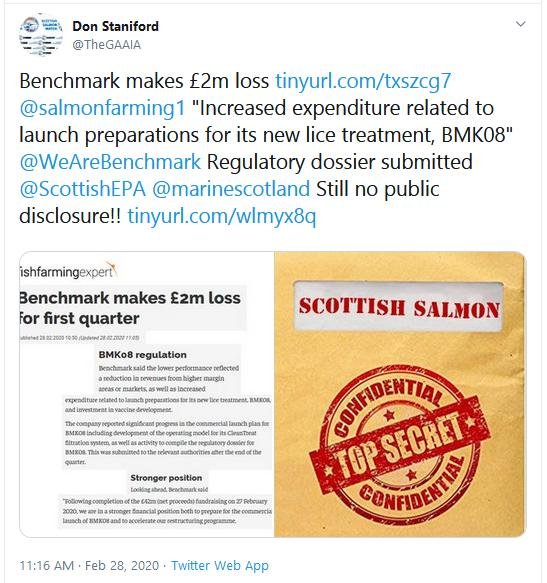
- Further to Scottish Salmon Watch's FOI request dated 20 May 2020 and letter dated 17 March 2020, could you please provide information on any scientific risk assessments of the ecosystem impacts of Imidacloprid (BMK08/Ectosan) in salmon farming?
- Scottish Salmon Watch's letter to SEPA dated 17 March 2020 included the following (as yet unanswered) questions:
- What scientific research has SEPA conducted on the toxicity and environmental impact of Imidacloprid?
- Will SEPA be publishing a scientific dossier on environmental impacts of Imidacloprid (as SEPA has done so via Emamectin benzoate)?
- Will SEPA be conducting scientific studies and monitoring of impacts of Imidacloprid (as it has done so via Emamectin benzoate)?
- Will Bayer/Monsanto be making available to the public their 'Confidential' work on Imidacloprid (or will the chemical company behind Imidacloprid be taking the Merck behind the scenes route)?
Could the VMD and the Scottish Government (in addition to SEPA) please provide answers to the questions above?
- Could you please share any information you have detailing how Benchmark can magically make Imidicloprid disappear from wastewater effluent discharges?
- What environmental effects have been considered in relation to trials (whether proposed, aborted, in-progress or completed) in Scotland?
- What scientific research including ecological risk assessment has been conducted in relation to Imidacloprid remote discharges of effluent water in 'discharge zones'? At what concentration is Imidacloprid being measured down to?
- Has SEPA, VMD and/or the Scottish Government conducted any modelling of environmental impacts or any ecological risk assessments of Imidacloprid use in salmon farming? Please therefore provide information on any scientific risk assessments of the ecosystem impacts of Imidacloprid (BMK08/Ectosan) in salmon farming since June 2016.
Our Reply
As a general point, you should note that the Freedom of Information Act (FOIA) gives you an entitlement to information rather than documents and it is in this context that we have answered your request.
- Further to Scottish Salmon Watch's FOI request dated 20 May 2020 and letter dated 17 March 2020, could you please provide information on any scientific risk assessments of the ecosystem impacts of Imidacloprid (BMK08/Ectosan) in salmon farming?
We fully recognise and respect the obligations imposed by the FOIA on the VMD to act in a manner that is transparent and open in the public interest. However, the very real harm likely to ensue from the disclosure of the information you request warrants the balance to be taken in favour of non-disclosure.
We consider that the disclosure of the information would be likely to prejudice the commercial interests of Marketing Authorisation holders and that Section 43 applies here. Section 43 is subject to a public interest test balance. After careful consideration we have concluded that the public interest in withholding the information strongly outweighs that for disclosure in this case. Through the marketing authorisation system, the VMD receives information from commercial entities that constitutes trade secrets and information protected by patent, the publication of which would create significant commercial harm to the manufacturer. The commercial entities are required by law to provide the information and are put to great expense to generate it. In this case, the information you request could be used by other manufacturers to formulate, develop or market products that would compete with these products. We consider such factors weigh against the public interest in disclosing the information in question.
We also judge that Section 41 of the FOIA applies to this request. Section 41 is an absolute exemption and it is engaged in this case because the information carries the necessary quality of confidence. In this instance publishing such information could undermine the veterinary pharmaceuticals industry’s trust in the regulatory process as well as the companies’ commercial position. This could lead to companies becoming unwilling to place product on the UK market, which would have a detrimental impact on animal welfare. Further, the market as a whole benefits from establishing and upholding a robust process for marketing authorisations; this in turn requires appropriate protection of confidentiality, and the honouring of expectations of confidentiality.
- Scottish Salmon Watch's letter to SEPA dated 17 March 2020 included the following (as yet unanswered) questions:
- What scientific research has SEPA conducted on the toxicity and environmental impact of Imidacloprid?
- Will SEPA be publishing a scientific dossier on environmental impacts of Imidacloprid (as SEPA has done so via Emamectin benzoate)?
- Will SEPA be conducting scientific studies and monitoring of impacts of Imidacloprid (as it has done so via Emamectin benzoate)?
- Will Bayer/Monsanto be making available to the public their 'Confidential' work on Imidacloprid (or will the chemical company behind Imidacloprid be taking the Merck behind the scenes route)?
Please note that the VMD does not hold any recorded information in this area. The FOIA only gives you the right to request access to recorded information held by the VMD. The Information Commissioner’s Office guidance – “When is information covered by the Freedom of Information Act?”- is clear on this. It says the VMD is only required to provide information it already has in recorded form. Under the FOIA the VMD does not have to create new information or find the answer to a question.
- Could you please share any information you have detailing how Benchmark can magically make Imidicloprid disappear from wastewater effluent discharges?
Please note that the VMD does not hold any recorded information in this area. The FOIA only gives you the right to request access to recorded information held by the VMD. The Information Commissioner’s Office guidance – “When is information covered by the Freedom of Information Act?”- is clear on this. It says the VMD is only required to provide information it already has in recorded form. Under the FOIA the VMD does not have to create new information or find the answer to a question.
- What environmental effects have been considered in relation to trials (whether proposed, aborted, in-progress or completed) in Scotland?
Please note that the VMD does not hold any recorded information in this area. The FOIA only gives you the right to request access to recorded information held by the VMD. The Information Commissioner’s Office guidance – “When is information covered by the Freedom of Information Act?”- is clear on this. It says the VMD is only required to provide information it already has in recorded form. Under the FOIA the VMD does not have to create new information or find the answer to a question.
- What scientific research including ecological risk assessment has been conducted in relation to Imidacloprid remote discharges of effluent water in 'discharge zones'? At what concentration is Imidacloprid being measured down to?
We fully recognise and respect the obligations imposed by the FOIA on the VMD to act in a manner that is transparent and open in the public interest. However, the very real harm likely to ensue from the disclosure of the information you request warrants the balance to be taken in favour of non-disclosure.
We consider that the disclosure of the information would be likely to prejudice the commercial interests of Marketing Authorisation holders and that Section 43 applies here. Section 43 is subject to a public interest test balance. After careful consideration we have concluded that the public interest in withholding the information strongly outweighs that for disclosure in this case. Through the marketing authorisation system, the VMD receives information from commercial entities that constitutes trade secrets and information protected by patent, the publication of which would create significant commercial harm to the manufacturer. The commercial entities are required by law to provide the information and are put to great expense to generate it. In this case, the information you request could be used by other manufacturers to formulate, develop or market products that would compete with these products. We consider such factors weigh against the public interest in disclosing the information in question.
We also judge that Section 41 of the FOIA applies to this request. Section 41 is an absolute exemption and it is engaged in this case because the information carries the necessary quality of confidence. In this instance publishing such information could undermine the veterinary pharmaceuticals industry’s trust in the regulatory process as well as the companies’ commercial position. This could lead to companies becoming unwilling to place product on the UK market, which would have a detrimental impact on animal welfare. Further, the market as a whole benefits from establishing and upholding a robust process for marketing authorisations; this in turn requires appropriate protection of confidentiality, and the honouring of expectations of confidentiality.
- Has SEPA, VMD and/or the Scottish Government conducted any modelling of environmental impacts or any ecological risk assessments of Imidacloprid use in salmon farming? Please therefore provide information on any scientific risk assessments of the ecosystem impacts of Imidacloprid (BMK08/Ectosan) in salmon farming since June 2016.
We fully recognise and respect the obligations imposed by the FOIA on the VMD to act in a manner that is transparent and open in the public interest. However, the very real harm likely to ensue from the disclosure of the information you request warrants the balance to be taken in favour of non-disclosure.
We consider that the disclosure of the information would be likely to prejudice the commercial interests of Marketing Authorisation holders and that Section 43 applies here. Section 43 is subject to a public interest test balance. After careful consideration we have concluded that the public interest in withholding the information strongly outweighs that for disclosure in this case. Through the marketing authorisation system, the VMD receives information from commercial entities that constitutes trade secrets and information protected by patent, the publication of which would create significant commercial harm to the manufacturer. The commercial entities are required by law to provide the information and are put to great expense to generate it. In this case, the information you request could be used by other manufacturers to formulate, develop or market products that would compete with these products. We consider such factors weigh against the public interest in disclosing the information in question.
We also judge that Section 41 of the FOIA applies to this request. Section 41 is an absolute exemption and it is engaged in this case because the information carries the necessary quality of confidence. In this instance publishing such information could undermine the veterinary pharmaceuticals industry’s trust in the regulatory process as well as the companies’ commercial position. This could lead to companies becoming unwilling to place product on the UK market, which would have a detrimental impact on animal welfare. Further, the market as a whole benefits from establishing and upholding a robust process for marketing authorisations; this in turn requires appropriate protection of confidentiality, and the honouring of expectations of confidentiality.
Information releasable to the public
In keeping with the spirit and effect of the FOIA and the government’s Transparency Agenda, we may place this request on GOV.UK, in due course. We will not place information identifying you on the GOV.UK website.
Copyright
The information supplied to you continues to be protected by copyright. You are free to use it for your own purposes, including for private study and non-commercial research, and for any other purpose authorised by an exception in current copyright law. Documents (except photographs) can be also used in the UK without requiring permission for the purposes of news reporting. Any other re-use, for example commercial publication, would require the permission of the copyright holder.
Most documents produced by Defra will be protected by Crown Copyright. Most Crown copyright information can be re-used under the Open Government Licence. For information about the OGL and about re-using Crown Copyright information please see The National Archives website.
Copyright in other documents may rest with a third party. For information about obtaining permission from a third party see the Intellectual Property Office’s website.
Our Service
If you are unhappy with the service you have received in relation to your request and wish to make a complaint, you may request an internal review within two calendar months of the date of this e-mail. If you would like to request an internal review please write to the VMD via ati@vmd.gov.uk. If you are not content with the outcome of the internal review you have the right to apply directly to the Information Commissioner for a decision. The Information Commissioner can be contacted at:
Information Commissioner's Office
Wycliffe House
Water Lane
Wilmslow
Cheshire
SK9 5AF
Many thanks
Andrew Saunders
Freedom of Information Officer
Secret Salmon – VMD refuse FOI on the use of Imidacloprid in salmon farming: "We consider that the disclosure of the information would be likely to prejudice the commercial interests of Marketing Authorisation holders" @vmdgovuk https://t.co/pZU7MnuCX3 @WeAreBenchmark @FerdOwner pic.twitter.com/gwpxpkdciT
— Don Staniford (@TheGAAIA) July 8, 2020