FOI reveals Environment Canada knew Ectosan (BMK08) was the banned neonicotinoid in May 2019 but @WeAreBenchmark were "not willing to send the information on their technology" @environmentca @miljodir @Legemiddelinfo @NFdep @Folketrygdfond @GovCanHealth https://t.co/Jo2DOhVTD6 pic.twitter.com/sfO2CrOHEz
— Don Staniford (@TheGAAIA) June 25, 2021
Following a Freedom of Information request to the Norwegian Government (16 June 2021), here's a reply from the Norwegian Environment Agency dated 24 June 2021:
From: Inger Marie Haaland <inger.marie.haaland@miljodir.no>
Date: Thu, Jun 24, 2021 at 2:12 PM
Subject: Sv: FOI re. Benchmark's CleanTreat & Ectosan/BMK08/Imidacloprid since 1 January 2016 (Innsynsrett & Miljøinformasjonsloven)
To: salmonfarmingkills@gmail.com <salmonfarmingkills@gmail.com>
Cc: Innsyn <innsyn@miljodir.no>
Dear Mr. Staniford
We refer to your request regarding documents related to Benchmark and Ectosan, sent June the 16th.
The Norwegian Environment Agency has not been in contact with Benchmark. We have not received an application or made any decisions according to the environmental law for Benchmark and the use of Ectosan.
Benchmarks and Ectosan has been mentioned in email correspondence between the Norwegian Environment Agency and the Norwegian Food Safety Authority (Mattilsynet), the Norwegian Medicines Agency (Statens legemiddelverk), the County governor in Vestland and Environment and Climate Change Canada. These emails are included. Some of the information in these emails are restricted and are therefore censored (Censored text is marked). Restricted information regarding Ectosans active ingredient, the Celantreat technology and testing in Norway is described by the media and is therefore not censored (Cleantreat, Uttesting av nytt lusemiddel skaper støy i UK – FFE-redaktør intervjuet på BBC – Kyst.no, Vellykkede storskala feltforsøk med nytt lusemiddel – Kyst.no)
The collaboration group on delousing pharmaceuticals decided in their meeting on October the 25th 2018 to schedule a meeting with Benchmark early in 2019. However, no meeting with Benchmark was held. (Members of the collaboration group were the Directorate of Fisheries, the Norwegian Food Safety Authority and the Norwegian Environment Agency. The group was shut down in 2019).
Regards
Inger Marie Haaland
Senior Adviser, Marine management and industries
|
Norwegian Environment Agency Phone: +47 73 58 05 00 |
www.environmentagency.no – www.environment.no
The FOI disclosure by the Norwegian Environment Agency on 24 June 2021 included:
The Norwegian Food Safety Agency @InfoMattilsynet knew in April 2019 that Ectosan (re-named BMK08) was the banned neonicotinoid Imidacloprid @WeAreBenchmark @FerdOwner @HansBrattskar @NFdep @fiskeridir @miljodir @Legemiddelinfo
How much Norwegian salmon has been embalmed? pic.twitter.com/LQcy6FkAKk— Don Staniford (@TheGAAIA) June 25, 2021
Have Leroy or Scottish Sea Farms used the banned neonicotinoid Imidacloprid on salmon farms in Norway or Scotland? @LeroySeafood @scotseafarms @WeAreBenchmark @FerdOwner @HansBrattskar @NFdep @fiskeridir @InfoMattilsynet @miljodir @Legemiddelinfo @seafoodnorway @NorwayMFA pic.twitter.com/KfMyDsSwHu
— Don Staniford (@TheGAAIA) June 25, 2021
Here's Scottish Salmon Watch's FOI request filed on 16 June 2021 with the Norwegian Government's Directorate of Fisheries (similar FOI requests were also sent to other Norwegian Government agencies as well as salmon farming companies including Mowi, Leroy, SalMar and Grieg Seafood – read online here):
From: Don Staniford <salmonfarmingkills@gmail.com>
Date: Wed, Jun 16, 2021 at 12:36 PM
Subject: Fwd: FOI re. Benchmark's CleanTreat & Ectosan/BMK08/Imidacloprid since 1 January 2016 (Innsynsrett & Miljøinformasjonsloven)
To: Postmottak <postmottak@fiskeridir.no>
Please consider this a formal request for Information and request for environmental information under the relevant Norwegian legislation.
Please provide information on Benchmark's CleanTreat water purification system including field trials and any commercial use of Imidacloprid (marketed as Ectosan in 2017 and then re-named BMK08 in 2019 before being revealed in March 2020 as the toxic neonicotinoid Imidacloprid – prior to 2017 it could have been called D10-Aquatic Blast or another secret trade name) since 1 January 2016.
A FOI request dated 20 March 2020 to various Norwegian Government agencies cited a news report in Fish Farmer magazine in February 2020 detailing how "over the last 24 months, more than 35,000 tonnes of salmon in five Norwegian farms have been treated with BMK08". Read article in full online via https://www.pressreader.com/uk/fish-farmer/20200211/282488595733006
Please therefore include in your FOI reply any documentation (e.g. emails, letters and other information) relating to the use – via field trials and other methods – of Imidacloprid since 1 January 2016.
Please include any environmental risk assessments, residue studies, ecological surveys, benthic impact research, water quality testing and any other environmental impact information even if heavily redacted.
Please include any results of field trials, testing, assessment and any other information submitted by Benchmark, Mowi, Leroy and other salmon farming companies.
Please include information on any discussions with the European Medicines Agency, the Scottish Government, Benchmark, Mowi and other salmon farming companies in relation to the use of CleanTreat and Imidacloprid (BMK08/Ectosan – or any other name used by Benchmark or another company) since 1 January 2016.
Please include any discussions with respect to an MRL (Maximum Residue Limit) of Imidacloprid in the flesh of farmed salmon and the recent European Parliamentary opposition.
This would certainly include the two clinical trials referenced via https://www.fiskeridir.no/Akvakultur/Tildeling-og-tillatelser/Saertillatelser/Forskningstillatelser/Alle-forskningstillatelsene
The above projects involving D-10 Aquatic Blast (Imidacloprid) were reported in the media in August 2020 via:
https://www.hblad.no/2020/nyheter/tjente-25-mill-i-fjor/
As context to this FOI request, Scottish Salmon Watch understands that Norway is subject to the legal requirements of FOI and Environmental Information law: https://ojs.imodev.org/index.php/RIGO/article/view/10/71
However, last year Scottish Salmon Watch filed a FOI request with the Norwegian Government which was not replied to; namely:
FOI request on Imidacloprid to Norwegian Government & Norwegian Food Safety Authority (17 March 2020)
When Scottish Salmon Watch pressed the Norwegian Medicines Agency the response was that the information was "considered to be confidential":
From: Tonje Høy <tonje.hoy@legemiddelverket.no>
Date: Wed, Apr 29, 2020 at 3:58 PM
Subject: SV: Imidacloprid clinical trials approved by Norwegian Medicines Agency?
To: salmonfarmingkills@gmail.com <salmonfarmingkills@gmail.com>
Dear Mr. Staniford,
Information about clinical trials with veterinary medicinal products, performed by pharmaceutical industry, is considered to be confidential information. According to the Norwegian act on medicinal products §30 the NoMA is not allowed to disclose information that is considered commercially sensitive. We can therefore not disclose whether permission to perform clinical trials with imidacloprid has been granted.
Right of appeal
You can appeal against this decision within 3 weeks after you have received this e-mail. The right to appeal is founded on the Public Administration Act, chapter 6 (VI).
An appeal should be sent to The Norwegian Medicines Authority. If we do not take the appeal into account, the appeal is forwarded to the Ministry of Health and Care Services.
Your appeal can be put forward as an e-mail/letter or using the form found on www.legemiddelverket.no. Choose the English version, and then the link “Appeal against an Administrative decision”.
Kind regards
Tonje Høy, DVM, PhD
Scientific Director, Veterinary Medicine
Telephone: (+47) 922 80 497
www.noma.no
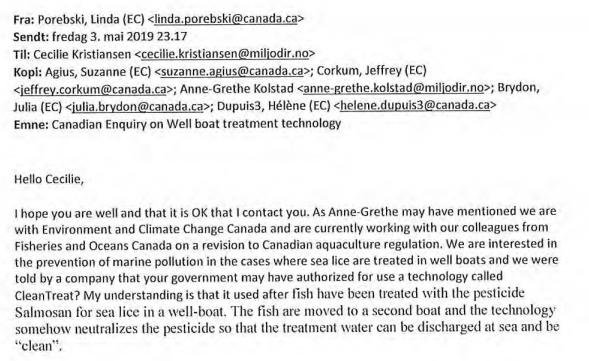
However, the Scottish Government and the Scottish Environment Protection Agency have been forced to disclose "commercially confidential" information on CleanTreat and Imidacloprid (Ectosan/BMK08); namely:
https://www.gov.scot/publications/foi-19-01398/
https://www.itspublicknowledge.info/202000275.aspx
A FOI disclosure by the Scottish Environment Protection Agency to Scottish Salmon Watch on 7 June 2021 included:
Minutes of a meeting between Benchmark, the Scottish Government, SEPA and the Veterinary Medicines Directorate in April 2019:
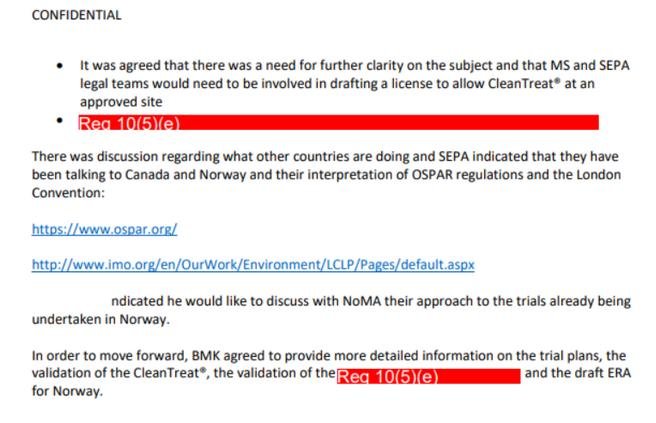
Here's another email obtained from SEPA via FOI on 7 June 2021 which references field trials in Norway:
Another email disclosed by SEPA via FOI on 7 June 2021 includes a timeline in relation to the use of CleanTreat with Ectosan (Imidacloprid/BMK08) since permission was granted for the first field trial in Norway in November 2016:
The above email states that the Norwegian field trials of Imidacloprid "were permitted in Norway with a level of discharge that appears to be above the EQS" [Environmental Quality Standard] which Scottish Salmon Watch suspects may breach EU law.
In any case, Scottish Salmon Watch is therefore asking for transparency and clarification from the Norwegian Government in relation to Imidacloprid use in salmon farming.
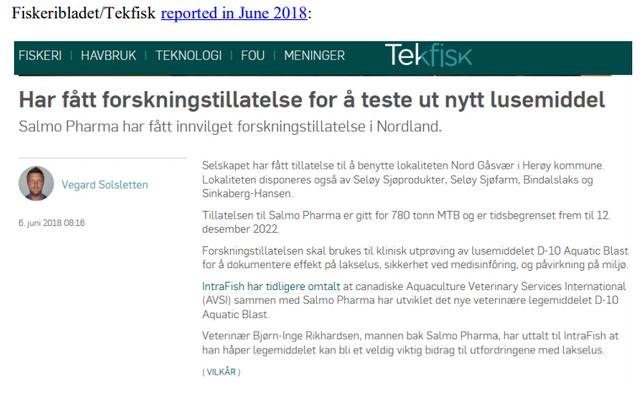
It is known via the trade press in 2016 that Salmo Pharma may have been the Norwegian company first involved in trialling Imidacloprid (perhaps then called D10-Aquatic Blast rather than Ectosan or BMK08): https://www.kyst.no/article/dette-er-det-nye-lusemiddelet-salmo-pharma-skal-teste-ut/
This June 2016 article – cited in Scottish Salmon Watch's FOI request of 17 March 2020 to the Norwegian Government – appears to identify companies involved in the first field trials of Imidacloprid in Norway:
Intrafish also appeared to report similar information via https://www.intrafish.no/nyheter/soker-to-fou-tillatelser-for-nytt-lusemiddel/1-1-766165
Apologies since I do not speak Norwegian and hence it is difficult to be sure. Hopefully your FOI reply can clarify things.
Via an 'Interpretation Request' from February 2019 published by the Aquaculture Stewardship Council, we also know that Mowi Norway may have been involved in a field trial of Imidacloprid in Norway: https://www.asc-aqua.org/what-you-can-do/get-certified/variance-request-interpretation-platform/QA0066/
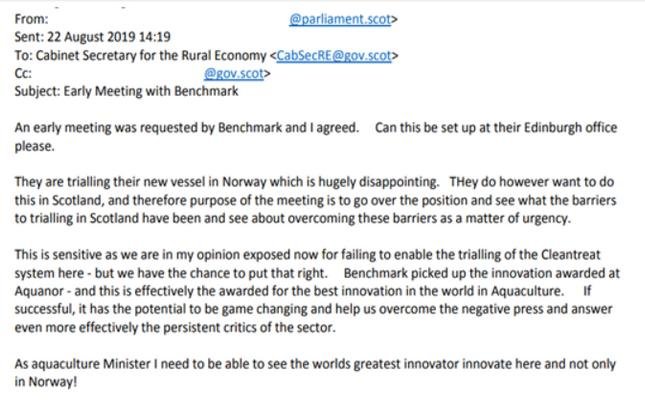
The owners of Scottish Sea Farms (Leroy and SalMar) may also be involved in field trials of Imidacloprid in Norway. A FOI disclosure by the Scottish Government in May 2021 included:
The Ferret reported last month (21 May 2021): "We are currently focused on launching our new sea lice solution, BMK08 and CleanTreat, in Norway".
You should be aware of recent opposition by the European Parliament to the use of Imidacloprid in salmon farming – including specific mention of the lack of transparency:
Intrafish reported (13 June 2021): "The commercial launch remains subject to marketing authorization in Norway. Though Norway is not a member of the EU, it is part of the European Medicines Agency, and follows its approval processes closely"
Please provide a receipt/acknowledgment for this FOI request.
Please provide the information electronically.
If you need clarification please do not hesitate to contact me.
Takk,
Don Staniford
Director, Scottish Salmon Watch
Which salmon farms in Norway are being permitted by @fiskeridir to use the banned neonicotinoid Imidacloprid (D-10 Aquatic Blast)? https://t.co/sojgslu2Wk @seafoodnorway @UK_NSC @NFdep @fiskeribladet @IntraFishNorge @WeAreBenchmark @FerdOwner @NorwaySeafoodUS@Fiskeoppdrett pic.twitter.com/gO4Ge4JDZw
— Don Staniford (@TheGAAIA) June 23, 2021
The Norwegian Medicines Agency replied (25 June 2021):
From: Tonje Høy <tonje.hoy@legemiddelverket.no>
Date: Fri, Jun 25, 2021 at 1:57 PM
Subject: Imidacloprid
To: Don Staniford <salmonfarmingkills@gmail.com>
To Mr. Don Staniford,
We refer to your e-mails of June 15th and June 17th 2021 requesting information regarding imidacloprid in Norwegian aquaculture.
No marketing authorisation for any veterinary medicinal product containing imidacloprid, and intended for treatment of sea lice infestations, has been granted in Norway.
The only legal use of the substance for sea lice treatment would therefore be in clinical trials. As previously stated, information on clinical trials cannot be disclosed as such information is considered business sensitive and therefore exempt from public disclosure.
With regard to purification/cleansing systems, the NoMA is not the competent authority for approval of such methods/systems.
Right of appeal
You can appeal against this decision within 3 weeks after you have received this e-mail. The right to appeal is founded on the Public Administration Act, chapter 6 (VI).An appeal should be sent to The Norwegian Medicines Authority. If we do not take the appeal into account, the appeal is forwarded to the Ministry of Health and Care Services. Your appeal can be put forward as an e-mail/letter or using the form found on www.legemiddelverket.no. Choose the English version, and then the link “Appeal against an Administrative decision”.
Kind regards
Tonje Høy, DVM, PhD
Scientific Director, Veterinary Medicine
Telephone: (+47) 922 80 497
www.noma.no
Read more via:
- Interferencia: "Salmon Farming – Norwegian company seeks to commercialize in Chile a pesticide banned by the EU for being 'very toxic' to aquatic life"
- Anyone for Norwegian Salmon Marinaded in the Banned Neonicotinoid Imidacloprid (don't worry it's ASC-approved)?!
- "EU deals potential setback to Benchmark’s rollout of new sea lice treatment"
- Fish Farmer: "MEPs condemn new lice treatment: who's right" "
- "EU setback for novel lice treatment"
- Breaking News: Victory in Europe – Parliament votes against the use of the toxic neonicotinoid Imidacloprid in farmed salmon!
- Fish Farming Expert: "Novel lice treatment threatened by European Parliament vote"
- Wave of European Opposition to Benchmark's Imidacloprid (BMK08) – the tide turns against neonicotinoids in salmon pharming!
- European Opposition to Neonicotinoid Contamination of Farmed Salmon – is BMK08 (Imidacloprid) dead in the water?
- The Guardian: "‘Novichok for insects’ may be approved for Scottish fish farms"
- Who are the toxic team lurking behind Benchmark's BMK08 (Imidacloprid) in Scottish salmon pharming?
- The Ferret: "Bee-killing pesticide treatment for fish farms backed by Scottish Government"
- Cleaning Tox-Sick Scottish Salmon – Benchmark's magic trick making the banned neonicotinoid Imidacloprid disappear!
- Rumsfeldian Fish Philosophy – Things We Know We Know, Things We Now Know We Don't Know & Things We Don't Know We Don't Know About 'Scottish' Salmon!
- SEPA ordered by Commissioner to Disclose 'Commercially Confidential' Information on CleanTreat & BMK08 (Imidacloprid) by 7 June
- Warning: Neonicotinoid Imidacloprid Use in Salmon Farming "Clears Another Hurdle" With European MRL
- Warning: Imidacloprid Moves A Step Closer to Commercial Use in Salmon Farming
- Nasty Neonicotinoid Plagues Salmon Farming in 2021
- Toxic Neonicotinoid "Close to Commercial Launch" in Salmon Farming!
- New Science Dogs Approval of Neonicotinoid Imidacloprid for Scottish Salmon!
- Imidacloprid Use in Scottish Salmon Farming?
- Secret Salmon – On the Trail of Imidacloprid Use in Scotland!
- British Wildlife: "Neonics in Salmon Farming – Alarm Bells Are Ringing"
- Caveat Chile – toxic neonicotinoid Imidacloprid (BMK08) coming soon!
- Toxic Politics – Meet the Scottish Government's Minister for Sinister Chemicals
- Imidacloprid Licensed for Use by Mowi in Loch Ailort?
- Further Scientific Opposition to Imidacloprid Use in Salmon Farming
- Patent lifts lid on neonicotinoid use in salmon farming
- Horrible History: Imidacloprid Use in Salmon Farming
- Scientific Opposition to Neonicotinoid Use in Salmon Farming
- Backgrounder: "Imidacloprid – A Neonicotinoid Insecticide Toxic to Aquatic Life"
- BBC Farming Today on "Neonicotinoids in Salmon Farming"
- Sunday Times: "Outlawed insecticide may be used on Scottish salmon"
- FOI letter on Scientific Concerns re. Imidacloprid Use in Scottish Salmon Farming
- Scottish Greens "Deeply Concerned" at Mowi's Field Trial of Neonicotinoid Imidacloprid in Loch Ailort
- Letter to the Prince of Wales re. bee-killing Imidacloprid use at royal warrant holder Mowi
- Secret Trials: 'Royal' Salmon Doused with Bee-Killing Insecticide Imidacloprid?
- Follow the Salmoney – the Norwegian tobacco billionaire behind the Neonicotinoid Imidacloprid
- ENDS Report: "Calls for greater transparency over ‘toxic’ new fish farm product"
- Letter to SEPA: Please Come Clean on Imidacloprid!
- Letter to Scottish Ministers, SEPA & the VMD re. Imidacloprid
- FOI requests on Imidacloprid to SEPA, VMD & the Scottish Government
- Revealed: Toxic Neonicotinoid Insecticide Used to 'CleanTreat' Lousy Salmon
- Please Come Clean on BMK08!
- Sunday Times: "Official fears revealed over toxic threat of salmon trade"
- Cleaning Tox-Sick Scottish Salmon
- CleanTreat FOI Disclosures by the Scottish Government to Scottish Salmon Watch
- Media Backgrounder: Chemically Embalmed Scottish Salmon
- All is Not Well With Sick Scottish Salmon
Anyone for Norwegian Salmon Marindaded in Banned Neonicotinoid Imidacloprid (don't worry it's ASC-approved)?! https://t.co/fn6TWu9qo4#BMK08 #Ectosan #D10AquaticBlast @WeAreBenchmark @FerdOwner @ASC_aqua
Is toxic Norwegian salmon reaching consumers? @seafoodnorway @REMA1000 pic.twitter.com/BpytMop4U2— Don Staniford (@TheGAAIA) June 17, 2021
Addendum:
Date: Fri, Jun 25, 2021 at 9:34 AM
Subject: Imidacloprid use on salmon farms in Norway?
To: Christine Børnes <christine.bornes@mattilsynet.no>
Cc: Inger Marie Haaland <inger.marie.haaland@miljodir.no>
From: Line Ruden <Line.Ruden@mattilsynet.no>
Date: Thu, Jul 1, 2021 at 1:55 PM
Subject: RE: FOI re. Benchmark's CleanTreat & Ectosan/BMK08/Imidacloprid since 1 January 2016 (Innsynsrett & Miljøinformasjonsloven)
To: salmonfarmingkills@gmail.com <salmonfarmingkills@gmail.com>
Dear Don Staniford,
Thank you for your request for information. Most of your questions fall however under The Norwegian Medicines Agency’s administrative area.
We do not have any documentation relating to the use of imidacloprid. Environmental risk assessment, residue studies, ecological surveys, benthic impact research, water quality testing and other environmental impact information belong to The Norwegian Medicines Agency’s area of responsibility. It is also them that approves the clinical trials. We have not had any discussions with the European Medicines Agency, the Scottish Government, Benchmark, Mowi or other salmon farming companies in relation to the use of CleanTreat and imidacloprid.
However, we do have a proposal for consultation with regard to imidacloprid. We propose a change in the legislation regulating the maximum residue limit in foodstuffs of animal origin, with regard to imidacloprid. You can view the proposal at our web page: Endringer i forskrift om grenseverdier for legemiddelrester i næringsmidler fra dyr (MRL-forskriften) | Mattilsynet. The Commission implementing regulation is in English.
Best regards,
Line Ruden
Senior Adviser/Toxicologist
Norwegian Food Safety Authority, Regulations and Control Department, Fish health and welfare
Tel: +47 22 77 85 87
Location: Stensberggata 27, Oslo
Address: Head Office, Felles postmottak, P.O.Box 383, N-2381 Brumunddal
www.mattilsynet.no www.matportalen.no